Tomocube which posted its first ever quarterly operating profit in the fourth quarter of last year also appeared to benefit from rising expectations for improved earnings in the first quarter of this year.
In contrast VUNO’s stock price declined after the company failed to obtain U.S. Food and Drug Administration (FDA) 510(k) clearance for VUNO Med DeepCARS its AI powered medical device designed to predict cardiac arrest.
|
Lunit Secures Financial Stability, Accelerates Growth of Lunit SCOPE
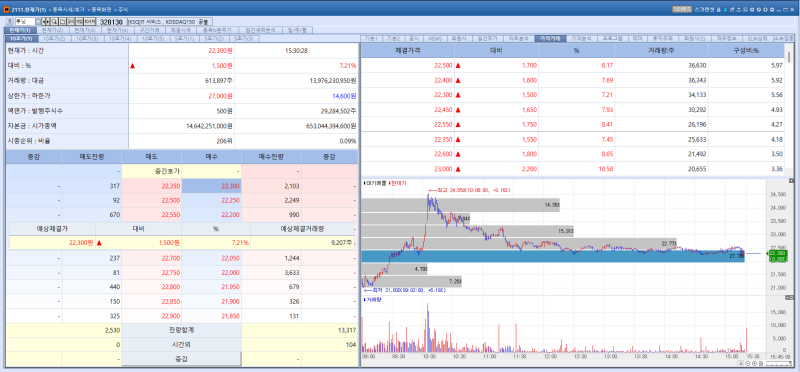
According to KG Zeroin’s MP DOCTOR (formerly MarketPoint), Lunit shares rose 7.21% from the previous trading day to close at 22,300 won on the day. The gain was attributed to the continued effect of the exrights adjustment following the company’s bonus issue on May 4. The reference price was set at 20,750 won.
In a bonus issue, the number of outstanding shares increases while the company’s market capitalization remains unchanged, prompting the Korea Exchange to artificially lower the stock price on the exrights date.
This often creates the perception that the stock has become relatively cheaper, which can drive share prices higher. Earlier, Lunit announced a rights offering worth 211.5 billion won. Separately, the company is carrying out a one-for-one bonus issue for shareholders and plans to complete the listing of new shares on May 26.
Through the 211.5 billion won capital increase, Lunit aims to strengthen its financial stability while accelerating growth and improving profitability. The company is pinning its hopes on the continued expansion of Lunit SCOPE, its medical artificial intelligence (AI) solution.
Last year Lunit SCOPE surpassed 10 billion won in annual sales for the first time more than doubling from the previous year. Since its initial launch in 2023 the platform has maintained annual growth of over 100% for three consecutive years.
Lunit SCOPE is an AI biomarker platform that precisely analyzes the tumor microenvironment (TME), including immune cells in cancer patients, using only hematoxylin and eosin (H&E) slide images. Its key competitive edge lies in significantly reducing both cost and processing time compared with conventional molecular diagnostics, while also improving accuracy and reproducibility.
Lunit expects Lunit SCOPE to achieve more than double growth this year, as the platform is increasingly being selected by global pharmaceutical companies as a partner for drug development rather than merely being used for research purposes. The company is targeting a turnaround to operating profit on an EBITDA basis by the end of this year.
A Lunit official said, “The stock price appears to have reflected expectations for improved earnings driven by the ex rights effect from the bonus issue and the growth potential of Lunit SCOPE.”
|
Tomocube, Which Achieved Its First Ever Quarterly Profit Last Year Expected to Post Stronger Earnings Growth This Year
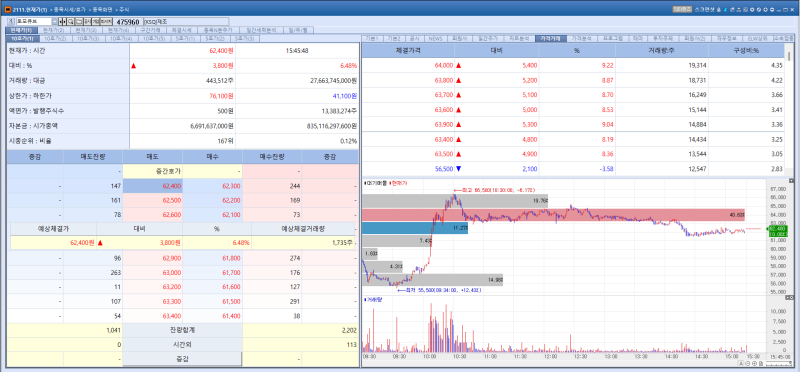
Tomocube shares rose 6.48% from the previous trading day to close at 62,400 won. In the fourth quarter of last year, the company posted sales of 5 billion won and an operating profit of 400 million won, achieving its first quarterly profit since its establishment. Revenue surged 449% compared with the same period a year earlier. With 44% of its annual sales concentrated in the fourth quarter alone, Tomocube was seen as having demonstrated strong order competitiveness among pharmaceutical companies and research institutions.
The company’s earnings growth this year is expected to be driven by wider global adoption of its flagship HT-X1 device and the launch of its next generation model, the HT-X1 Max. Tomocube has been recognized for reshaping the cell analysis market with its innovative technology.
Conventional cell analysis methods have relied on chemical staining, which often causes phototoxicity from fluorescent signals, leading to concerns over cell damage and deformation.
In particular, conventional two dimensional (2D) cell analysis has faced limitations in accurately identifying the complex three dimensional (3D) structures of cells. In contrast, Tomocube’s holotomography technology is considered the only method capable of observing intracellular organelles without labeling through precise refractive index measurements while also enabling long term analysis of thick cell tissues without staining.
Tomocube is targeting annual sales of 100 billion won by 2028. Last year, the company reported revenue of 11.3 billion won and an operating loss of 5.6 billion won.
A Tomocube official said “We have already exceeded the figures presented in our securities registration statement. If the current growth trend continues, achieving our target is not impossible.”
|
VUNO Fails to Obtain FDA Clearance for VUNO Med DeepCARS Plans to Reapply
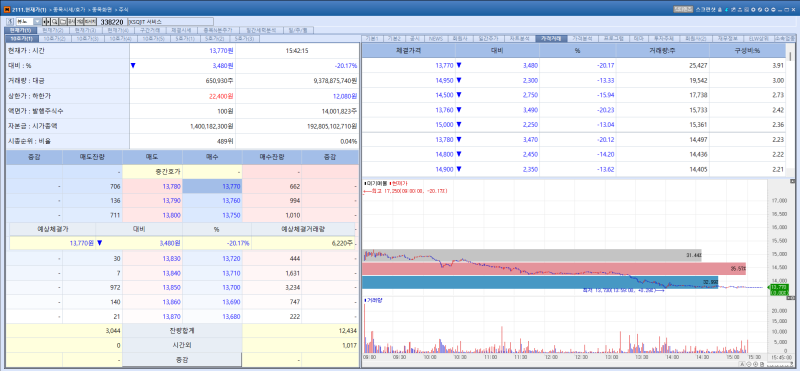
VUNO shares closed at 13,770 won on the day, down 20.17% from the previous trading session. The decline followed news that the company failed to obtain U.S. Food and Drug Administration (FDA) 510(k) clearance for VUNO Med-DeepCARS. VUNO plans to reorganize its clinical data and reapply for certification.
VUNO CEO Lee Ye ha said through the company’s website the previous day that the firm had been notified by the FDA on April 30 (U.S. time) that VUNO Med-DeepCARS had received a “Not Substantially Equivalent” (NSE) determination regarding its 510(k) submission.
FDA 510(k) clearance is a representative approval process required for entry into the U.S. medical device market. The system evaluates the safety and performance of new devices by comparing their substantial equivalence with already approved products. VUNO submitted its 510(k) application to the FDA in early 2023.
In June of the same year, the company became the first Korean medical AI firm to receive the FDA’s Breakthrough Device designation. Although VUNO had expected approval within 2024, the review process was delayed due to requests for supplementary materials before the company recently received the NSE notice.
Lee said “The FDA’s decision does not negate the core technology or clinical value of DeepCARS. We interpret it as a request for additional evidence, such as proof of substantial equivalence in the intended use environment, compared with existing products.”
He added “We feel a heavy sense of responsibility for not meeting the high standards required by U.S. regulators. At the same time, we were able to clearly confirm the clinical standards and expectations demanded by the U.S. market. VUNO plans to reorganize its clinical data and promptly reapply for certification.”

.png)