Koh Young hit the daily upper limit after posting preliminary first-quarter results that exceeded market expectations. ViGenCell attracted investor interest as the first beneficiary under Korea’s Advanced Regenerative Bio Act. ToolGen edged up on expectations for a license-out deal after PharmEdaily premium content was made freely available on a portal.
|
Koh Young beats expectations in Q1… brain surgery robot sales still pending
According to KG Zeroin’s MP Doctor (formerly MarketPoint), Koh Young closed at 42,300 won, up 29.95% (9,750 won) from the previous day. The company had disclosed its first quarter earnings outlook the day before, which is understood to have surpassed market expectations.
Koh Young reported first-quarter revenue of 72.7 billion won, up 42.3% from 51.1 billion won a year earlier. Operating profit reached 9.8 billion won, more than tripling from 3.1 billion won, while net profit rose nearly fivefold to 15.6 billion won from 3.2 billion won.
According to Edaily’s reporting, most of the first-quarter revenue came from 3D solder paste inspection (SPI) and 3D automated optical inspection (AOI) equipment.
A company official said, “Quarterly performance exceeded market expectations. Although the first quarter is typically a slow season and we did not actively communicate with the market, rising sales from AI server customers and increased AI solution revenue had a positive impact.”
He added “As for surgical robots, rapid growth has yet to materialize. We have exported one unit to the U.S. so far received approval in Japan this January, and are awaiting sales there. We also filed for regulatory approval in China this April.”
Koh Young’s brain surgery robot “Geniant Cranial” obtained FDA 510(k) clearance in January last year and approval from Japan’s Ministry of Health, Labour and Welfare (MHLW) this January, enabling immediate sales in both markets. However, growth has been slower than expected as the company is pursuing direct sales rather than using distributors.
The official said, “In the early stage, we are operating under a direct sales model to strengthen technical support and initial setup rather than using distributors. We expect the medical robot business to contribute to revenue and are focusing on overseas expansion.”
|
ViGenCell becomes first case of ‘treatment plan approval’ under Advanced Regenerative Bio Act
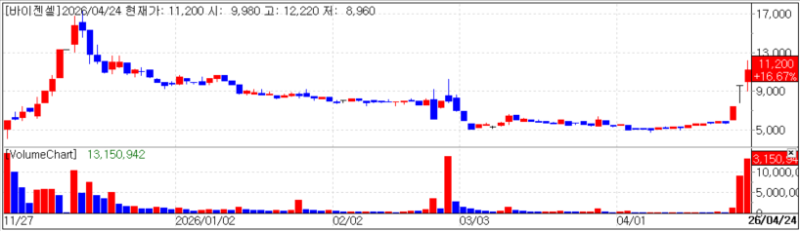
ViGenCell closed at 11,200 won, up 16.67% (1,600 won) from the previous day. The company announced that its autologous immune cell therapy “VT-EBV-N” received approval for its treatment plan from the Ministry of Health and Welfare’s Advanced Regenerative Medicine and Advanced Biopharmaceutical Review Committee for patients in complete remission of EBV-positive extranodal NK/T-cell lymphoma (a type of blood cancer) with a high risk of recurrence.
This approval is fundamentally different from regulatory authorization by the Ministry of Food and Drug Safety (MFDS). It is understood to be the first case under a new treatment approval track established following amendments to the Advanced Regenerative Bio Act that took effect in February last year. It is reported as Korea’s first “treatment plan approval” case.
In a call with Edaily, ViGenCell Vice President Shin Young-bae said, “As far as we know, all other applicants failed. Since this is the first case, the Ministry is handling it very carefully. It can be understood as a system that allows earlier use of therapies for patients.”
ViGenCell plans to work closely with medical institutions to provide treatment opportunities and pursue conditional approval and license-out of VT-EBV-N.
Meanwhile Boryung transferred its shares to Gaeun Global for 8 billion won in January 2025, relinquishing its position as the largest shareholder. Currently, Gaeun Global holds 2.19 million shares (10.68%) and Boryung holds 2.188 million shares (10.68%), making them the largest and second-largest shareholders, respectively, by a narrow margin.
Gaeun Global appears to have invested with expectations of synergy between ViGenCell and Therabest, an unlisted induced pluripotent stem cell (iPSC)-based drug developer in which it holds a 43.93% stake.
Gaeun Global was founded by psychiatrist Ki Pyeong seok, director of Gaeun Hospital. He currently serves as CEO of both ViGenCell and Therabest, overseeing management as well as clinical and R&D operations. Gaeun Global is led by CEO Ki Young wook son of Ki Pyeong seok who holds a 49.79% stake.
|
ToolGen pushes commercialization of ‘commercial crop’ business
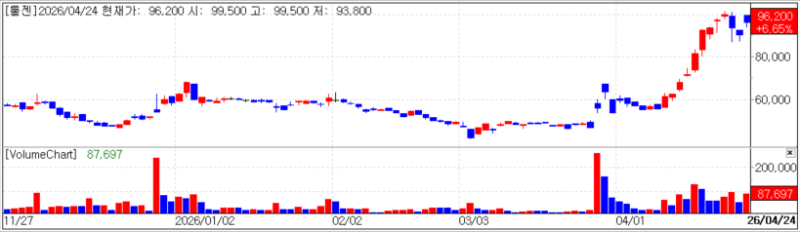
ToolGen closed at 96,200 won, up 6.65% (6,000 won) from the previous day. The gain is attributed to the release of PharmEdaily premium content on a portal the same day titled “ToolGen CEO Yoo Jong-sang Targeting seed business license-out this year… drought-tolerant pepper deal taking shape.”
According to the report, ToolGen has overhauled its seed business, which had previously faced delays, and is accelerating efforts toward a license out within the year. After restructuring its pipeline with a focus on profitability and commercialization potential, negotiations with global companies led by drought-tolerant peppers have gained momentum.
In a recent interview with Edaily at the company’s headquarters in Gangseo gu, Seoul, CEO Yoo Jong-sang said, “We are currently in discussions with global companies for technology transfer involving key crops, including peppers, and aim to sign contracts within the year. Negotiations for peppers, in particular, are quite advanced.”
ToolGen has fundamentally changed its seed business strategy. In the past, it built a broad pipeline across various crops based on its gene-editing technology, but many projects failed to reach commercialization.
In June last year, the company recruited Sung Dong ryeol formerly of global seed giant Monsanto, as head of its seed business division. Yoo explained, “Previously, the pipeline was spread horizontally rather than vertically, and with multiple attempts running in parallel, we lacked focus. Vice President Sung has taken the lead in streamlining the portfolio and reorganizing it around projects with strong marketability and profitability.”
Unlike drug development, the seed business has the advantage of faster commercialization due to fewer clinical trial and regulatory burdens. However, it also faces structural limitations, particularly restricted access to commercial seed varieties. Even if superior genotypes are identified in research strains, transferring those traits into commercially viable seeds owned by major companies is not easy.
Accordingly, ToolGen has restructured its development strategy to focus on crops with high potential or secured pathways for transition into commercial varieties. At the same time, it has narrowed its target traits to those in high market demand.
Yoo added “While our partners are global companies, we cannot disclose their names. However, since we have streamlined the pipeline to projects with high likelihood of technology transfer, we expect results much faster than before.”
He continued, “The seed business addresses structural demand driven by climate change and food security. We aim to secure early revenue by focusing on two key areas: drought tolerance and herbicide resistance.”


.png)
.png)







![[속보]코스피 6700 돌파…최고점 경신](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26042800565t.jpg)