Pipeline momentum draws investor attention
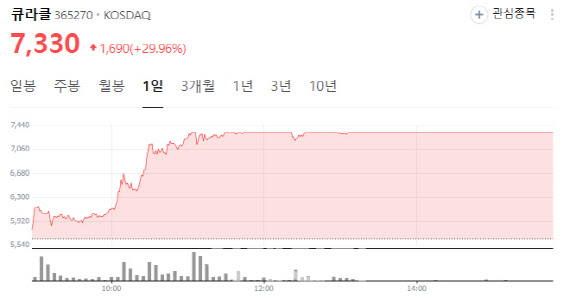
According to KG Zeroin MP Doctor (formerly MarketPoint), Curacle shares jumped 29.96% to close at 7,330 won, hitting the daily limit-up. The rally followed news that Curacle signed a strategic partnership with Boryung for the development of its diabetic nephropathy treatment candidate CU01, and a PharmEdaily report suggesting imminent technology export of MT-101 also boosted sentiment.
|
The PharmEdaily report, first released as a paid article on Sept. 10 and made free on Sept. 11 at 9:30 a.m., also fueled trading. It revealed that Curacle had signed multiple material transfer agreements (MTAs) with overseas pharmaceutical companies for its kidney disease candidate MT-101. The article compared Curacle to Olix, which rebounded after inking a deal with Eli Lilly following a pipeline setback, and suggested Curacle could follow a similar trajectory as “the next Olix.” Analysts noted that competition among multiple global drugmakers signals a high probability of a license-out deal within the year.
Powder product takes flight
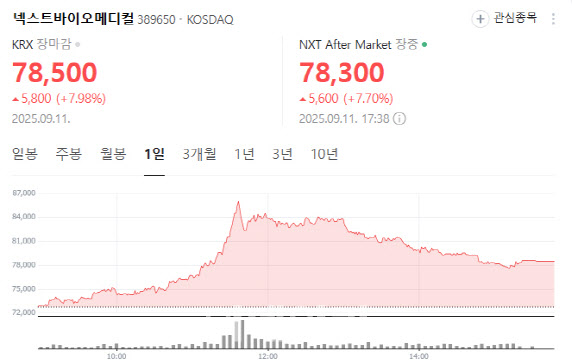
NextBioMedical rose 7.98% to 78,500 won. Investor optimism grew on news of strong exports for its endoscopic hemostatic spray powder NexPowder.
|
A company official said, “It is difficult to attribute the stock move to a single factor, but sales expansion in the U.S. and Japan and clinical data supporting the product’s efficacy appear to be driving market interest.”
NexPowder was designated for reimbursement under Japan’s national health insurance on Sept. 5, with sales to begin this month. That comes just six months after approval from Japan’s Pharmaceuticals and Medical Devices Agency (PMDA). The product had already secured clearance from the U.S. Food and Drug Administration, Europe’s CE-MDR (Medical Device Regulation), and Korea’s Ministry of Food and Drug Safety. Global distribution is handled by Medtronic, the world’s largest medical device maker.
Clinical data debate weighs on shares
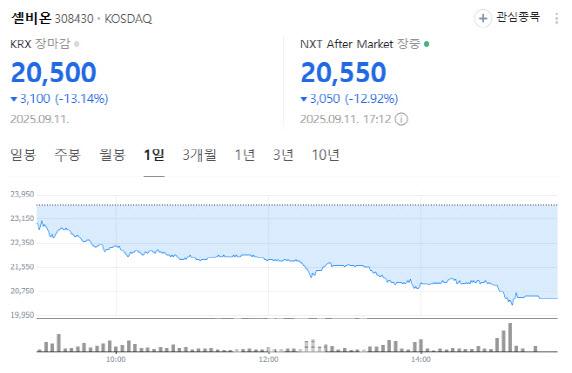
Cellbion fell 13.14% to 20,500 won. The decline followed ongoing questions about the interpretation of topline data from a Phase 2 trial of its prostate cancer radiopharmaceutical candidate 177Lu-pocuvotide (Pocuvotide).
|
Cellbion countered that differences stem from varying sample sizes between interim and final analyses, and that the final dataset, processed under regulatory standards, is the one relevant for review. It emphasized that Pocuvotide outperformed Pluvicto’s Phase 2 Asian trial results (ORR 54.4% vs. 30%) and showed a lower adverse event rate (57.1% vs. 93.3%).
After plunging 21% on Sept. 5, Cellbion shares had stabilized with small moves through Sept. 8~10, before sliding again on Sept. 11. Trading volume spiked to more than 1.13 million shares, underscoring heavy selling pressure.
The company plans to file for conditional approval with Korea’s regulator after receiving the final CSR in the fourth quarter. A Cellbion official said, “We apologize for the confusion caused during the clinical announcement and will continue to provide transparent and accurate information to maintain shareholder trust.”

.png)
.png)









![[그해오늘] 살인으로 끝난 '사령카페' 회원들의 인연](https://image.edaily.co.kr/images/vision/files/NP/S/2026/04/PS26043000002t.jpg)