Inventage Lab limit-up on Boehringer MTA… Quratis, G2GBIO up
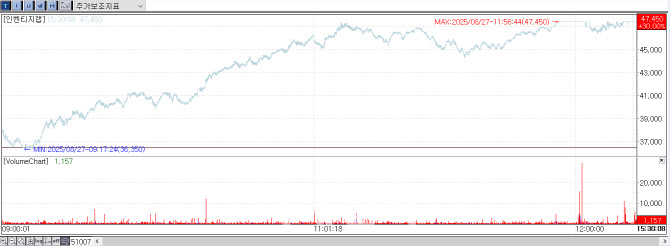
According to KG Zeroin MP Doctor (formerly Market Point), Inventage Lab closed at 47,450 won, up 10,950 won (30%) from the previous day. The company’s share price began to soar at 10:31 a.m., reaching the upper limit at 11:56 a.m. Although it briefly eased, the stock maintained its ceiling from 12:17 p.m. through the market close.
|
Industry observers expect the agreement to be related to Boehringer Ingelheim’s next-generation GLP-1 obesity therapy currently in clinical development. Since September last year, the two companies have been co-developing a peptide-based obesity treatment by applying Boehringer Ingelheim’s candidate to Inventage Lab’s drug delivery system (DDS) platform, IVL-DrugFluidic. Under the collaboration, Boehringer is focusing on efficacy and clinical studies, while Inventage Lab handles formulation and process development. The IVL-DrugFluidic platform can extend dosing intervals from one month up to 12 months, depending on the drug.
Inventage Lab has also been working to establish a production base in Europe, signing an MOU with a European pharma company that owns an EU GMP-certified facility. The company is also in detailed discussions on licensing out its manufacturing platform technology to a European partner.
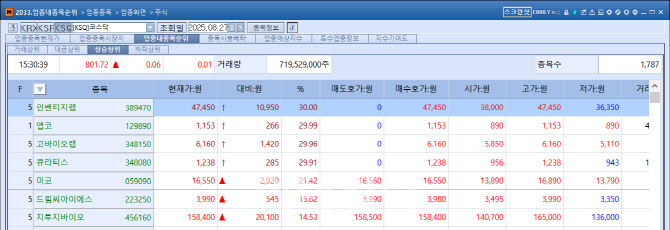
Amid this momentum, investor attention turned to Quratis, which Inventage Lab acquired earlier this year to utilize its GMP facilities. Quratis shares also hit the upper limit, closing at 1,238 won, up 285 won (29.91%) from the previous session after a sharp rise starting around 11:30 a.m.
An Inventage Lab official commented, “The news that our joint development agreement with Boehringer Ingelheim was expanded into an MTA and that we are securing European production facilities appears to have driven the stock higher. In Quratis’s case, we are equipping facilities to enable clinical sample production and early commercialization using our platform technology.”
Meanwhile, G2GBIO, which owns the long-acting drug delivery platform InnoLAMP, also rallied strongly from 10:31 a.m. The stock closed at 158,400 won, up 20,100 won (14.53%) from the previous day. The surge was attributed to investor focus on the fact that G2GBIO is also collaborating with Boehringer Ingelheim.
|
KoBioLabs hit the ceiling after announcing that its strain Akkermansia muciniphila KBL983 for obesity treatment was granted a U.S. patent. Its shares soared from 10 a.m., closing at the upper limit of 6,160 won, up 1,420 won (29.96%).
The news drew attention because the strain has potential for developing an oral microbiome-based obesity drug. Derived from the human microbiome, KBL983 has been shown to induce GLP-1 expression and increase brown fat activity. This supports the development of an oral therapeutic for weight control, impaired glucose tolerance/diabetes, atherosclerosis, fatty liver, and other obesity-related metabolic diseases.
Notably, obesity drugs based on KBL983 are expected to avoid common side effects of existing GLP-1 receptor agonists, such as rebound weight gain and gastrointestinal discomfort, while offering the convenience of oral administration.
A KoBioLabs official stated, “We are advancing multiple pipelines, including treatments for autism spectrum disorder, metabolic diseases, and inflammatory bowel disease. Among them is the obesity therapy. Preparing patents has been part of our commercialization strategy, and the U.S. patent registration of KBL983 is a significant step forward.”
Alteogen slips 4.4% on KOSPI transfer delay concerns
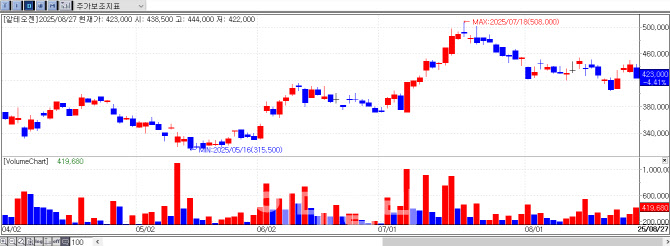
Alteogen, the largest company on the KOSDAQ by market cap, dropped sharply from the opening, closing at 423,000 won, down 19,500 won (4.41%). The decline was attributed to disappointment over the growing perception that a KOSPI transfer this year is unlikely, prompting some selling.
|
In response to this rapid mobilization, CEO Park Soon-jae issued a public notice on the 27th, stating, “We will pursue the KOSPI transfer as early as possible, with internal preparations underway.” He added, “Even if the timing differs from what shareholders expect, we intend to ensure a well-prepared transfer. We ask for patience.” Park hinted that the formal transfer process may not begin until 2026.
Following this, some retail shareholders who had joined the campaign announced they would no longer engage in collective action. This sparked debate among investors―some argued there was no need for further activism now that the CEO had officially confirmed the transfer, while others insisted the process should begin immediately.
One retail shareholder remarked, “Even if an extraordinary shareholders’ meeting were held and the process started right away, the earliest the transfer could be completed would be Q2 next year. By then, Keytruda SC will already have gained traction in the market―so why delay?”
An industry insider commented, “The CEO’s statement shows they want to proceed cautiously. For long-term investors, this isn’t necessarily a negative.”

.png)







!['개과천선' 한국판 패리스 힐튼 서인영의 아파트[누구집]](https://image.edaily.co.kr/images/vision/files/NP/S/2026/05/PS26050300075t.jpg)


