“Pipeline nearing results … providing opportunity to existing shareholders”
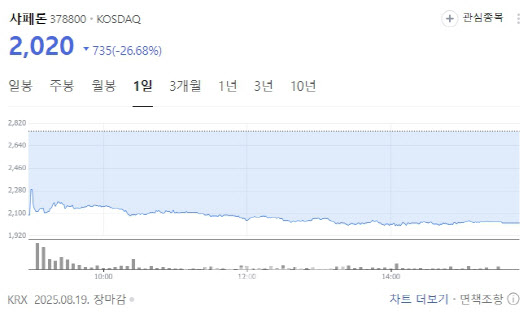
According to KG Zeroin MP Doctor (formerly MarketPoint), Shaperon closed at 2,020 won, down 26.68% from the previous session.
Shaperon said on Aug. 18 it will raise 30 billion won ($22.2 million) through a rights offering followed by a public subscription of forfeited shares. The planned issue price is 1,866 won per share, with 16.1 million new shares to be allocated. Any forfeited shares will be fully underwritten by securities firms.
|
Chronic losses and the absence of out-licensing deals remain key issues. Since listing, the company has posted annual net losses of 10 billion to 15 billion won without establishing a stable revenue base. Of two licensing agreements signed in the past, neither has resulted in commercialization; one deal with Bridge Biotherapeutics for a pulmonary fibrosis therapy was terminated last year.
This is the second straight year Shaperon has pursued a rights offering. In 2023, it targeted 35 billion won but managed to raise only about 12.7 billion won due to weak investor demand.
Asked why it chose a rights issue again, Shaperon told Edaily, “With commercialization of major pipelines such as our atopic dermatitis therapy approaching, we judged it best to give existing shareholders the first opportunity to participate.”
The firm plans to use proceeds for global Phase 2 trials in atopic dermatitis, Phase 1 studies in Alzheimer’s and pulmonary fibrosis, alopecia treatment development, nanobody immuno-oncology research, and new healthcare businesses.
“Accelerating clinical and commercialization efforts”
Corestem ChemOn ended at 2,065 won, down 25.45%. The company disclosed on Aug. 18 it will raise about 37.8 billion won ($28 million) through a rights offering with a public sale of forfeited shares.
In a notice, Corestem ChemOn said large-scale funding was needed to commercialize its ALS stem cell therapy Neuronata-R and strengthen its balance sheet. Of the total proceeds, 12.5 billion won will go toward clinical trials and R&D, while 24.3 billion won will be used to repay exchangeable bonds and loans.
Yang Gil-an, CEO of Corestem ChemOn, said, “This rights issue marks a turning point for the company’s future growth. With wise execution, it will allow us to deliver greater results. We will focus all resources on accelerating clinical trials and commercialization of our core pipeline.”
Neuronata-R has completed Phase 3 trials in both Korea and the United States. In April, the company submitted a request for label expansion to Korea’s Ministry of Food and Drug Safety (MFDS). On Aug. 19, the firm also announced that the MFDS had notified it of an upcoming Good Clinical Practice (GCP) inspection in September. Corestem ChemOn said the review was proceeding smoothly and that it was preparing thoroughly for both domestic and global approvals.
The company plans to hold a pre-BLA (biologics license application) meeting with the U.S. Food and Drug Administration in the second half of this year.
Shares fall despite easing of risks
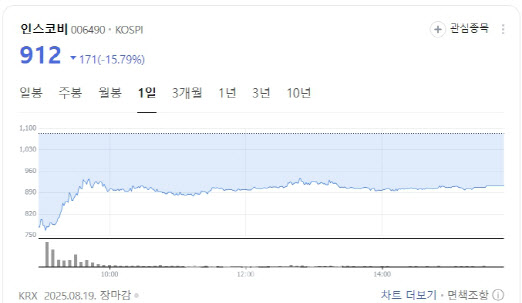
Incosbi closed at 912 won, down 15.79%. The company said its subsidiary Cellumed had reduced litigation damages from 24 billion won to 16.5 billion won, easing some uncertainty. Still, investors appeared unconvinced, citing doubts over whether planned asset sales would translate into growth. Adding to concerns, Incosbi recently received a “disclaimer of opinion” from its auditor on its semiannual report.
|
To secure liquidity, Incosbi is pushing the sale of key assets, including a 120MW data center in Busan and its nanosilicon business. The company says the sale could cover litigation payments while also funding new business expansion.
In addition, Incosbi is seeking to strengthen its global biotech business through its U.S. unit, Apimeds Pharmaceuticals US (APUS). Established to advance Phase 3 trials of autoimmune therapy Apitox, APUS raised $13.5 million in May via an NYSE American listing. Apitox, derived from bee venom, is Korea’s first approved natural-substance drug and the nation’s sixth approved new drug overall.
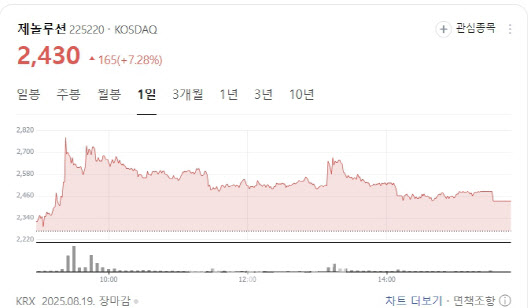
Patent filing lifts Genolution shares
Genolution shares rose 7.28% to 2,430 won. The firm said it had filed a domestic patent for a DNA methylation biomarker that predicts progression of precancerous cervical lesions, lifting investor sentiment.
|
Cervical cancer generally has a good prognosis when detected early, but current screening methods cannot reliably identify high-risk patients. Analysts said the new biomarker could reduce unnecessary follow-up tests and overtreatment while shifting the paradigm toward personalized prevention and management.
A Genolution official, commenting on the day’s share price gain, said, “We understand that today’s announcement of our patent filing for a DNA methylation biomarker predicting cervical cancer progression has drawn considerable interest. However, stock price movements are determined by market conditions and investor decisions, so it is difficult to directly link them to a single factor.”
The official added, “This research marks an important first step in expanding Genolution’s R&D beyond infectious disease diagnostics into cancer precision diagnostics. Cervical cancer is an area where early prediction and prevention can significantly contribute to patient outcomes and reduce social costs. Based on the discovered biomarker, we plan to actively pursue clinical validation and expand global partnerships.”

.png)
.png)